
To precisely measure a wave's energy would take an infinite amount of time while measuring a wave's exact instance in space would require to be collapsed onto a single moment which would have indefinite energy. Later, in 1927, a German physicist called Werner Heisenberg proposed that it is not possible to know where exactly an electron is located when it behaves like a wave and what its velocity is simultaneously because by trying to take any measurements, we would be disturbing it in some way. Very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa. Heisenberg Uncertainty Principle Definition. You could do the same thought experiment with energy and time. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. 
There is likewise a minimum for the product of the uncertainties of the energy and time. There is a minimum for the product of the uncertainties of these two measurements. Trying to pin a thing down to one definite position will make its momentum less well pinned down, and vice-versa. The position and momentum of a particle cannot be simultaneously measured with arbitrarily high precision. Furthermore, it shows that there is a definite relationship to how well each can be known relative to the other. Werner Heisenberg stumbled on a secret of the universe: Nothing has a definite position, a definite trajectory, or a definite momentum. The Heisenberg Uncertainty Principle Equation is the mathematical expression of the fact that the position and velocity of a particle cannot be known simultaneously. His most well-known thought experiment involved photographing an electron. (c) the necessity of using all of them at different moments for a comprehensive account of the totality of phenomena that we must consider. Heisenberg sometimes explained the uncertainty principle as a problem of making measurements. (b) the possibility of applying each one of them separately at any given point and. Similarly, a wave with a perfectly measurable momentum has a wavelength that oscillates over all space infinitely and therefore has an indefinite position. The Uncertainty principle is also called the Heisenberg uncertainty principle. (a) a mutual exclusivity of certain phenomena, entities, or conceptions and yet. A wave that has a perfectly measurable position is collapsed onto a single point with an indefinite wavelength and therefore indefinite momentum according to de Broglie's equation. Heisenbergs uncertainty principle is a very precise mathematical statement about the nature of a quantum system. Let's consider if quantum variables could be measured exactly. In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) 1 is any of a variety of mathematical inequalities 2 asserting a fundamental limit to the accuracy the values for certain related pairs of physical quantities of a particle, such as position, x, and. 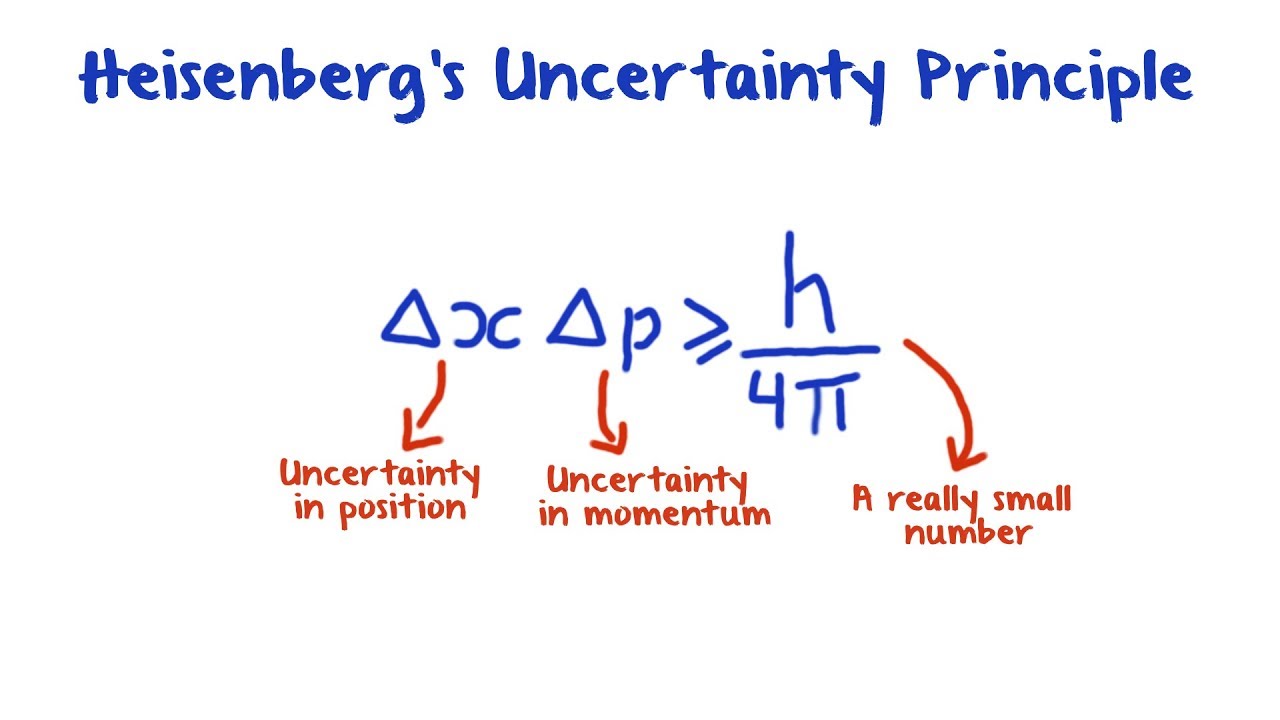
Though widely repeated in textbooks, this physical argument is now known to be fundamentally misleading.\) : A wave packet in space Uncertainty principle of Heisenberg, 1927. Explore the Heisenberg uncertainty principle by calculating uncertainty in position given the uncertainty in momentum for Bohr model of hydrogen. This ascribes the uncertainty in the measurable quantities to the jolt-like disturbance triggered by the act of observation. The Heisenberg uncertainty principle states that there is a limit to how precisely certain pairs of physical properties of a particle can be known simultaneously. The original heuristic argument that such a limit should exist was given by Heisenberg, after whom it is sometimes named the Heisenberg principle. Imagine driving a car fitted with a GPS navigation system that glitches. The formal inequality relating the standard deviation of position σx and the standard deviation of momentum σp was derived by Earle Hesse Kennard later that year and by Hermann Weyl in 1928. The Heisenberg uncertainty principle says that we cannot know both the position and the momentum of a particle at once. Origin of Heisenberg Uncertainty Principle Named for Werner Heisenberg, its discoverer, translated into English from a variety of original German terms. For instance, in 1927, Werner Heisenberg stated that the more precisely the position of some particle is determined, the less precisely its momentum can be known, and vice versa. Heisenberg Uncertainty Principle definition: Uncertainty principle. Uncertainty principle In quantum mechanics, the uncertainty principle is any of a variety of mathematical inequalities asserting a fundamental limit to the precision with which certain pairs of physical properties of a particle known as complementary variables, such as position x and momentum p, can be known simultaneously.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
